The Science
This is the science underpinning our work, but without giving away our key innovations, of course.
If you’re interested in working with us and need to know more, please get in touch and we’ll be happy to talk.
The Energy-Efficiency Curve
How to make a DPI’s performance independent of inspiratory effort
One of the primary aims of any respiratory drug delivery system is to deliver a known quantity of drug, independently of how the patient inhales. Another is to minimise the amount of drug that lands in the mouth and throat rather than reaching where it’s supposed to go: the lung. However, DPIs are notoriously sensitive to how the patient inhales, and deposit most of the drug in the mouth and throat. But there is a way of achieving both of our aims with the same approach.
The powder within a Dry Powder Inhaler (DPI) is typically formed of tiny respirable particles of the drug itself (the Active Pharmaceutical Ingredient, or API) attached to larger, inactive ‘carrier’ particles, usually lactose. While still very small, due to their size the carrier particles typically land in the mouth and throat, whereas the API is small enough to be carried into the deep lung. When the patient inhales and the powder moves through the inhaler, a proportion of the API is knocked off the lactose particles and makes it to the deep lung. This proportion is called the Fine Particle Fraction (FPF), and is typically only 20-40% of the API – the rest lands in the mouth and throat with the lactose particles it is still attached to, and this can cause unpleasant side effects.
It therefore makes sense to try to minimise the amount of drug that lands in the mouth and throat, or put another way, to maximise the amount that separates from the carrier particles, i.e. to maximise the FPF. Not only does this minimise side effects, but also reduces drug wastage and cost.
But maximising the FPF has another benefit: it can make the performance of the inhaler insensitive to how the patient inhales, thereby delivering a more consistent dose.
Dry Powder Inhalers (DPIs) are known for being sensitive to how hard the patient inhales – the harder the patient inhales, the greater the FPF and the larger the dose they receive, and vice versa. The inevitable variation in inhalation effort results in a variation in the dose being delivered. For inhaled drugs that make it to market this might mean sub-optimal treatment; however this issue can easily result in promising new treatments never making it to market, as they fail clinical trials due to excessive dosing variability.
However if we look at the Energy-Efficiency curve, we can see that by maximising the FPF we can minimise its variation due to changes in inspiratory effort.
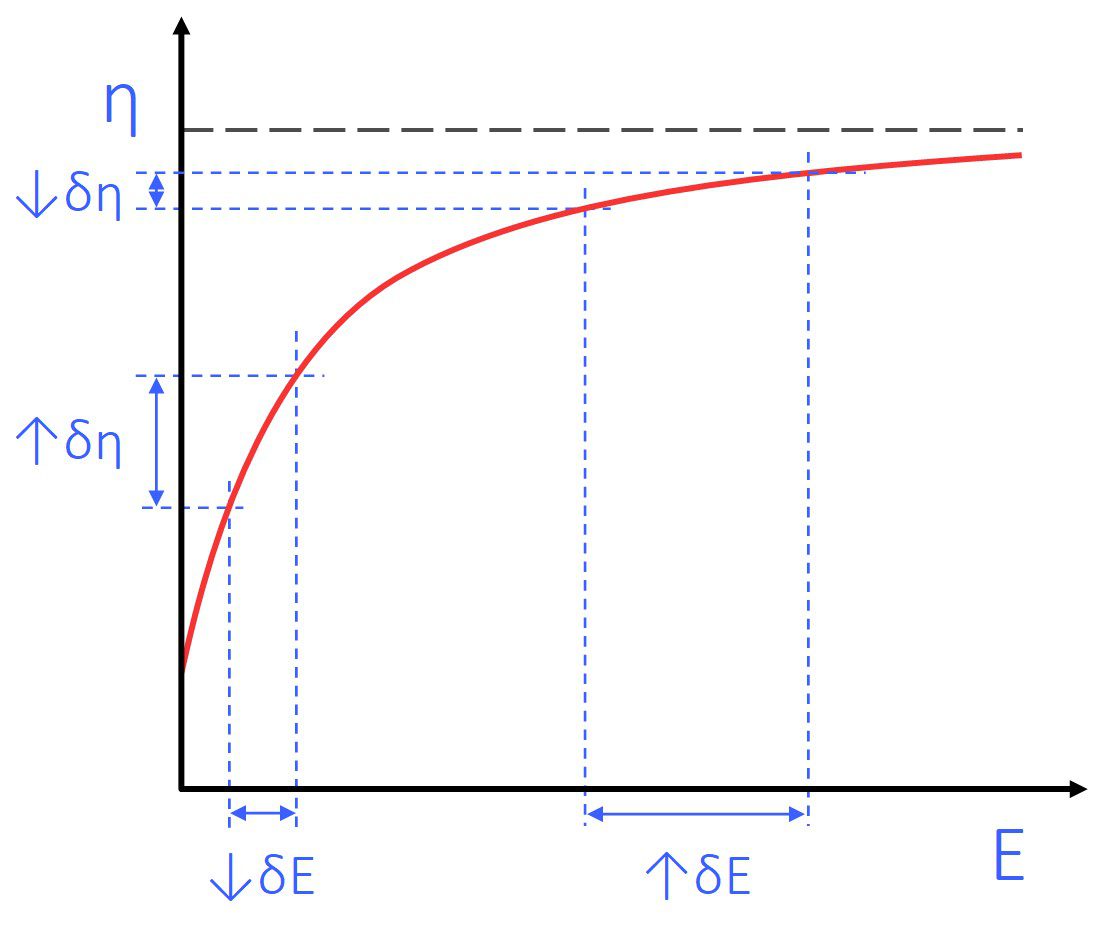

Figure 1: Schematic diagram showing the effect of input energy on Fine Particle Fraction for typical passive DPIs. We call this diagram the Energy-Efficiency curve, as it reflects how efficient the device is at aerosolising the API.
Dry Powder Inhalers get the energy to break up the powder and separate the API from the lactose from the inhalation effort. The relationship between Fine Particle Fraction and input energy (inhalation effort) is shown by the Energy-Efficiency curve (Figure 1). This curve can be considered as two parts – a steep section and a flatter element.
DPIs typically perform on the steep part of this curve.. This is a particularly suboptimal operating region, as even a small difference in the input energy (↓𝛿E) results in a large difference in the FPF (↑𝛿FPF).
As one of the primary aims is to deliver a known quantity of drug, independently of how the patient inhales, operating on the steepest part of the energy efficiency curve is unfortunately the worst place to be.
However, if the DPI were to operate on the flat part of the Efficiency-Energy curve, a large variation in input energy (↑𝛿E) causes minimal variation in FPF (↓𝛿FPF): i.e. the DPI’s performance is now largely independent of inhalation effort.
This is exactly what our αeolus technology is designed to do: by maximising the FPF, it both minimises the amount of drug that is deposited in the mouth and throat, and delivers a consistent dose that is independent of how hard the patient inhales.